Your Answer Is Incorrect Try Again How Many Sigma Bonds Are Present in the Following Compound
Learning Outcomes
- Explain the concept of diminutive orbital hybridization
- Determine the hybrid orbitals associated with various molecular geometries

Figure 1. The hypothetical overlap of 2 of the 2p orbitals on an oxygen atom (red) with the 1s orbitals of two hydrogen atoms (blue) would produce a bond bending of 90°. This is not consistent with experimental testify.
Thinking in terms of overlapping atomic orbitals is ane fashion for u.s.a. to explain how chemical bonds grade in diatomic molecules. However, to sympathise how molecules with more ii atoms grade stable bonds, we crave a more detailed model. As an example, permit us consider the water molecule, in which we have i oxygen atom bonding to two hydrogen atoms. Oxygen has the electron configuration 1south two2south 22p 4, with two unpaired electrons (one in each of the 2 2p orbitals). Valence bond theory would predict that the two O–H bonds form from the overlap of these two 2p orbitals with the anesouth orbitals of the hydrogen atoms. If this were the case, the bond angle would be xc°, every bit shown in Effigy one (note that orbitals may sometimes be drawn in an elongated "airship" shape rather than in a more realistic "plump" shape in social club to brand the geometry easier to visualize), considering p orbitals are perpendicular to each other.. Experimental show shows that the bond angle is 104.five°, not 90°. The prediction of the valence bond theory model does not match the existent-world observations of a water molecule; a different model is needed.
Quantum-mechanical calculations advise why the observed bail angles in H2O differ from those predicted past the overlap of the 1s orbital of the hydrogen atoms with the iip orbitals of the oxygen atom. The mathematical expression known equally the wave function, ψ, contains information nigh each orbital and the wavelike properties of electrons in an isolated atom. When atoms are jump together in a molecule, the wave functions combine to produce new mathematical descriptions that have unlike shapes. This process of combining the wave functions for atomic orbitals is called hybridization and is mathematically accomplished by the linear combination of atomic orbitals, LCAO, (a technique that we will encounter again later). The new orbitals that event are called hybrid orbitals. The valence orbitals in an isolated oxygen atom are a iis orbital and three twop orbitals. The valence orbitals in an oxygen cantlet in a h2o molecule differ; they consist of four equivalent hybrid orbitals that point approximately toward the corners of a tetrahedron (Effigy 2). Consequently, the overlap of the O and H orbitals should result in a tetrahedral bond angle (109.5°). The observed angle of 104.5° is experimental evidence for which quantum-mechanical calculations requite a useful explanation: Valence bond theory must include a hybridization component to requite accurate predictions.

Figure ii. (a) A water molecule has 4 regions of electron density, so VSEPR theory predicts a tetrahedral arrangement of hybrid orbitals. (b) 2 of the hybrid orbitals on oxygen comprise lone pairs, and the other 2 overlap with the 1s orbitals of hydrogen atoms to class the O–H bonds in H2O. This description is more consistent with the experimental structure.
The following ideas are important in understanding hybridization:
- Hybrid orbitals exercise not exist in isolated atoms. They are formed but in covalently bonded atoms.
- Hybrid orbitals accept shapes and orientations that are very unlike from those of the atomic orbitals in isolated atoms.
- A gear up of hybrid orbitals is generated by combining atomic orbitals. The number of hybrid orbitals in a ready is equal to the number of atomic orbitals that were combined to produce the set.
- All orbitals in a set of hybrid orbitals are equivalent in shape and free energy.
- The type of hybrid orbitals formed in a bonded atom depends on its electron-pair geometry as predicted by the VSEPR theory.
- Hybrid orbitals overlap to course σ bonds. Unhybridized orbitals overlap to form π bonds.
In the following sections, we shall talk over the common types of hybrid orbitals.
sp Hybridization
The beryllium atom in a gaseous BeCl2 molecule is an example of a central atom with no solitary pairs of electrons in a linear arrangement of three atoms. In that location are two regions of valence electron density in the BeCl2 molecule that correspond to the two covalent Be–Cl bonds. To adapt these two electron domains, 2 of the Be atom'south four valence orbitals will mix to yield 2 hybrid orbitals. This hybridization process involves mixing of the valence s orbital with i of the valence p orbitals to yield 2 equivalent sp hybrid orbitals that are oriented in a linear geometry (Figure three). In this figure, the set of sp orbitals appears similar in shape to the original p orbital, only there is an important difference. The number of atomic orbitals combined always equals the number of hybrid orbitals formed. The p orbital is i orbital that can concord upwards to two electrons. The sp set is two equivalent orbitals that point 180° from each other. The two electrons that were originally in the s orbital are now distributed to the two sp orbitals, which are one-half filled. In gaseous BeCl2, these half-filled hybrid orbitals will overlap with orbitals from the chlorine atoms to form two identical [latex]\sigma[/latex] bonds.

Effigy 3. Hybridization of an southward orbital (blueish) and a p orbital (crimson) of the same cantlet produces two sp hybrid orbitals (imperial). Each hybrid orbital is oriented primarily in just one direction. Annotation that each sp orbital contains one lobe that is significantly larger than the other. The set of two sp orbitals are oriented at 180°, which is consequent with the geometry for 2 domains.
Nosotros illustrate the electronic differences in an isolated Be atom and in the bonded Exist atom in the orbital energy-level diagram in Figure 4. These diagrams correspond each orbital by a horizontal line (indicating its energy) and each electron by an arrow. Free energy increases toward the peak of the diagram. Nosotros employ one upward arrow to indicate one electron in an orbital and two arrows (up and downward) to point two electrons of opposite spin.

Figure four. This orbital free energy-level diagram shows the sp hybridized orbitals on Be in the linear BeCl2 molecule. Each of the two sp hybrid orbitals holds one electron and is thus half filled and available for bonding via overlap with a Cl 3p orbital.
When atomic orbitals hybridize, the valence electrons occupy the newly created orbitals. The Exist atom had two valence electrons, and then each of the sp orbitals gets 1 of these electrons. Each of these electrons pairs up with the unpaired electron on a chlorine cantlet when a hybrid orbital and a chlorine orbital overlap during the germination of the Be–Cl bonds.
Any primal cantlet surrounded by merely 2 regions of valence electron density in a molecule will exhibit sp hybridization. Other examples include the mercury cantlet in the linear HgCltwo molecule, the zinc cantlet in Zn(CHiii)ii, which contains a linear C–Zn–C arrangement, and the carbon atoms in HCCH and COii.
Check out the University of Wisconsin-Oshkosh "What Are Hybrid Orbitals?" website to learn about visualizing hybrid orbitals in three dimensions.
sp 2 Hybridization
The valence orbitals of a central cantlet surrounded by three regions of electron density consist of a set of iii sp 2 hybrid orbitals and one unhybridized p orbital. This arrangement results from sp 2 hybridization, the mixing of 1 south orbital and two p orbitals to produce three identical hybrid orbitals oriented in a trigonal planar geometry (Effigy v).

Figure 5. The hybridization of an s orbital (bluish) and two p orbitals (ruby) produces 3 equivalent sp two hybridized orbitals (imperial) oriented at 120° with respect to each other. The remaining unhybridized p orbital is not shown here, but is located forth the z axis.

Figure 6. This alternating style of cartoon the trigonal planar sp 2 hybrid orbitals is sometimes used in more crowded figures.
Although breakthrough mechanics yields the "plump" orbital lobes as depicted in Figure 5, sometimes for clarity these orbitals are drawn thinner and without the minor lobes, as in Figure half dozen, to avoid obscuring other features of a given illustration.
We will utilise these "thinner" representations whenever the true view is too crowded to easily visualize.
The observed structure of the borane molecule, BH3, suggests sp 2 hybridization for boron in this chemical compound. The molecule is trigonal planar, and the boron atom is involved in 3 bonds to hydrogen atoms (Effigy 7).

Figure 7. BHiii is an electron-scarce molecule with a trigonal planar structure.
Nosotros can illustrate the comparison of orbitals and electron distribution in an isolated boron cantlet and in the bonded cantlet in BHiii equally shown in the orbital free energy level diagram in Figure eight. Nosotros redistribute the three valence electrons of the boron atom in the three sp 2 hybrid orbitals, and each boron electron pairs with a hydrogen electron when B–H bonds form.

Figure 8. In an isolated B cantlet, in that location are 1 2s and three 2p valence orbitals. When boron is in a molecule with three regions of electron density, three of the orbitals hybridize and create a prepare of three sp ii orbitals and 1 unhybridized iip orbital. The iii half-filled hybrid orbitals each overlap with an orbital from a hydrogen cantlet to form iii σ bonds in BHthree.
Whatsoever central cantlet surrounded by 3 regions of electron density will exhibit sp ii hybridization. This includes molecules with a lone pair on the fundamental atom, such as ClNO (Figure 9), or molecules with two single bonds and a double bond connected to the central atom, as in formaldehyde, CH2O, and ethene, H2CCH2.

Effigy 9. The cardinal atom(s) in each of the structures shown contain iii regions of electron density and are sp ii hybridized. Every bit nosotros know from the discussion of VSEPR theory, a region of electron density contains all of the electrons that point in 1 direction. A lonely pair, an unpaired electron, a single bond, or a multiple bail would each count as one region of electron density.
sp three Hybridization
The valence orbitals of an atom surrounded by a tetrahedral organisation of bonding pairs and solitary pairs consist of a set of iv sp 3 hybrid orbitals. The hybrids effect from the mixing of one s orbital and all three p orbitals that produces iv identical sp three hybrid orbitals (Figure 10). Each of these hybrid orbitals points toward a dissimilar corner of a tetrahedron.

Figure ten. The hybridization of an south orbital (bluish) and three p orbitals (cherry-red) produces four equivalent sp 3 hybridized orbitals (purple) oriented at 109.five° with respect to each other.
A molecule of methane, CH4, consists of a carbon cantlet surrounded by four hydrogen atoms at the corners of a tetrahedron. The carbon atom in methyl hydride exhibits sp iii hybridization. We illustrate the orbitals and electron distribution in an isolated carbon atom and in the bonded atom in CH4 in Figure 11. The four valence electrons of the carbon atom are distributed equally in the hybrid orbitals, and each carbon electron pairs with a hydrogen electron when the C–H bonds grade.

Effigy 11. The iv valence diminutive orbitals from an isolated carbon atom all hybridize when the carbon bonds in a molecule similar CH4 with iv regions of electron density. This creates four equivalent sp 3 hybridized orbitals. Overlap of each of the hybrid orbitals with a hydrogen orbital creates a C–H σ bond.
In a methane molecule, the 1southward orbital of each of the iv hydrogen atoms overlaps with one of the iv sp 3 orbitals of the carbon atom to form a sigma ([latex]\sigma[/latex]) bond. This results in the formation of iv strong, equivalent covalent bonds between the carbon atom and each of the hydrogen atoms to produce the methane molecule, CH4.
The structure of ethane, CtwoH6, is similar to that of marsh gas in that each carbon in ethane has iv neighboring atoms bundled at the corners of a tetrahedron—three hydrogen atoms and 1 carbon atom (Effigy 12). Nevertheless, in ethane an sp 3 orbital of one carbon atom overlaps end to cease with an sp 3 orbital of a second carbon atom to course a σ bond between the two carbon atoms. Each of the remaining sp iii hybrid orbitals overlaps with an due south orbital of a hydrogen cantlet to form carbon–hydrogen σ bonds. The construction and overall outline of the bonding orbitals of ethane are shown in Effigy 12. The orientation of the two CH3 groups is not fixed relative to each other. Experimental evidence shows that rotation around [latex]\sigma[/latex] bonds occurs hands.

Figure 12. (a) In the ethane molecule, C2H6, each carbon has four sp 3 orbitals. (b) These four orbitals overlap to form seven σ bonds.
An sp 3 hybrid orbital tin can also hold a lone pair of electrons. For example, the nitrogen atom in ammonia is surrounded past 3 bonding pairs and a lone pair of electrons directed to the four corners of a tetrahedron. The nitrogen atom is sp 3 hybridized with i hybrid orbital occupied by the lone pair. The molecular construction of water is consistent with a tetrahedral organisation of two lone pairs and two bonding pairs of electrons. Thus we say that the oxygen atom is sp iii hybridized, with 2 of the hybrid orbitals occupied by alone pairs and two by bonding pairs. Since alone pairs occupy more space than bonding pairs, structures that comprise alone pairs have bond angles slightly distorted from the ideal. Perfect tetrahedra have angles of 109.5°, but the observed angles in ammonia (107.3°) and h2o (104.5°) are slightly smaller. Other examples of sp 3 hybridization include CClfour, PCl3, and NClthree.
sp three d and sp 3 d two Hybridization
To describe the v bonding orbitals in a trigonal bipyramidal arrangement, we must utilize five of the valence shell diminutive orbitals (the due south orbital, the three p orbitals, and one of the d orbitals), which gives five sp 3 d hybrid orbitals. With an octahedral organisation of six hybrid orbitals, we must utilize vi valence beat diminutive orbitals (the southward orbital, the 3 p orbitals, and two of the d orbitals in its valence crush), which gives six sp iii d 2 hybrid orbitals. These hybridizations are only possible for atoms that have d orbitals in their valence subshells (that is, not those in the starting time or second menstruum).
In a molecule of phosphorus pentachloride, PCl5, there are 5 P–Cl bonds (thus five pairs of valence electrons around the phosphorus cantlet) directed toward the corners of a trigonal bipyramid. We use the 3s orbital, the three 3p orbitals, and i of the 3d orbitals to form the gear up of five sp three d hybrid orbitals (Figure 14) that are involved in the P–Cl bonds. Other atoms that showroom sp 3 d hybridization include the sulfur cantlet in [latex]\text{SF}_{four}[/latex] and the chlorine atoms in [latex]\text{ClF}_{3}[/latex] and in [latex]{\text{ClF}}_{4}^{\text{+}}.[/latex] (The electrons on fluorine atoms are omitted for clarity.)

Effigy 13. The three compounds pictured exhibit sp 3 d hybridization in the central atom and a trigonal bipyramid class. SF4 and ClF4 + have one lone pair of electrons on the primal atom, and ClFiii has 2 lone pairs giving it the T-shape shown.

Figure xiv. (a) The five regions of electron density around phosphorus in PClfive require five hybrid sp iii d orbitals. (b) These orbitals combine to course a trigonal bipyramidal construction with each big lobe of the hybrid orbital pointing at a vertex. Every bit earlier, at that place are likewise small lobes pointing in the reverse direction for each orbital (not shown for clarity).
The sulfur atom in sulfur hexafluoride, [latex]\text{SF}_{6}[/latex], exhibits sp 3 d 2 hybridization. A molecule of sulfur hexafluoride has six bonding pairs of electrons connecting six fluorine atoms to a single sulfur atom (Figure 15). In that location are no lone pairs of electrons on the central atom. To bond half-dozen fluorine atoms, the 3due south orbital, the three threep orbitals, and two of the iiid orbitals course six equivalent sp 3 d 2 hybrid orbitals, each directed toward a different corner of an octahedron. Other atoms that exhibit sp 3 d ii hybridization include the phosphorus atom in [latex]{\text{PCl}}_{6}^{-},[/latex] the iodine cantlet in the interhalogens [latex]{\text{IF}}_{6}^{\text{+}}[/latex], [latex]\text{IF}_{5}[/latex], [latex]{\text{ICl}}_{4}^{-}[/latex], [latex]{\text{IF}}_{4}^{-}[/latex] and the xenon atom in [latex]\text{XeF}_{iv}[/latex].

Figure 15. (a) Sulfur hexafluoride, SFvi, has an octahedral structure that requires sp iii d 2 hybridization. (b) The six sp 3 d 2 orbitals course an octahedral structure around sulfur. Over again, the pocket-size lobe of each orbital is non shown for clarity.
Assignment of Hybrid Orbitals to Central Atoms
The hybridization of an atom is adamant based on the number of regions of electron density that environs it. The geometrical arrangements characteristic of the various sets of hybrid orbitals are shown in Effigy 16. These arrangements are identical to those of the electron-pair geometries predicted by VSEPR theory. VSEPR theory predicts the shapes of molecules, and hybrid orbital theory provides an explanation for how those shapes are formed. To detect the hybridization of a primal atom, we tin use the post-obit guidelines:
- Decide the Lewis structure of the molecule.
- Make up one's mind the number of regions of electron density effectually an atom using VSEPR theory, in which single bonds, multiple bonds, radicals, and lone pairs each count as 1 region.
- Assign the set of hybridized orbitals from Figure 16 that corresponds to this geometry.
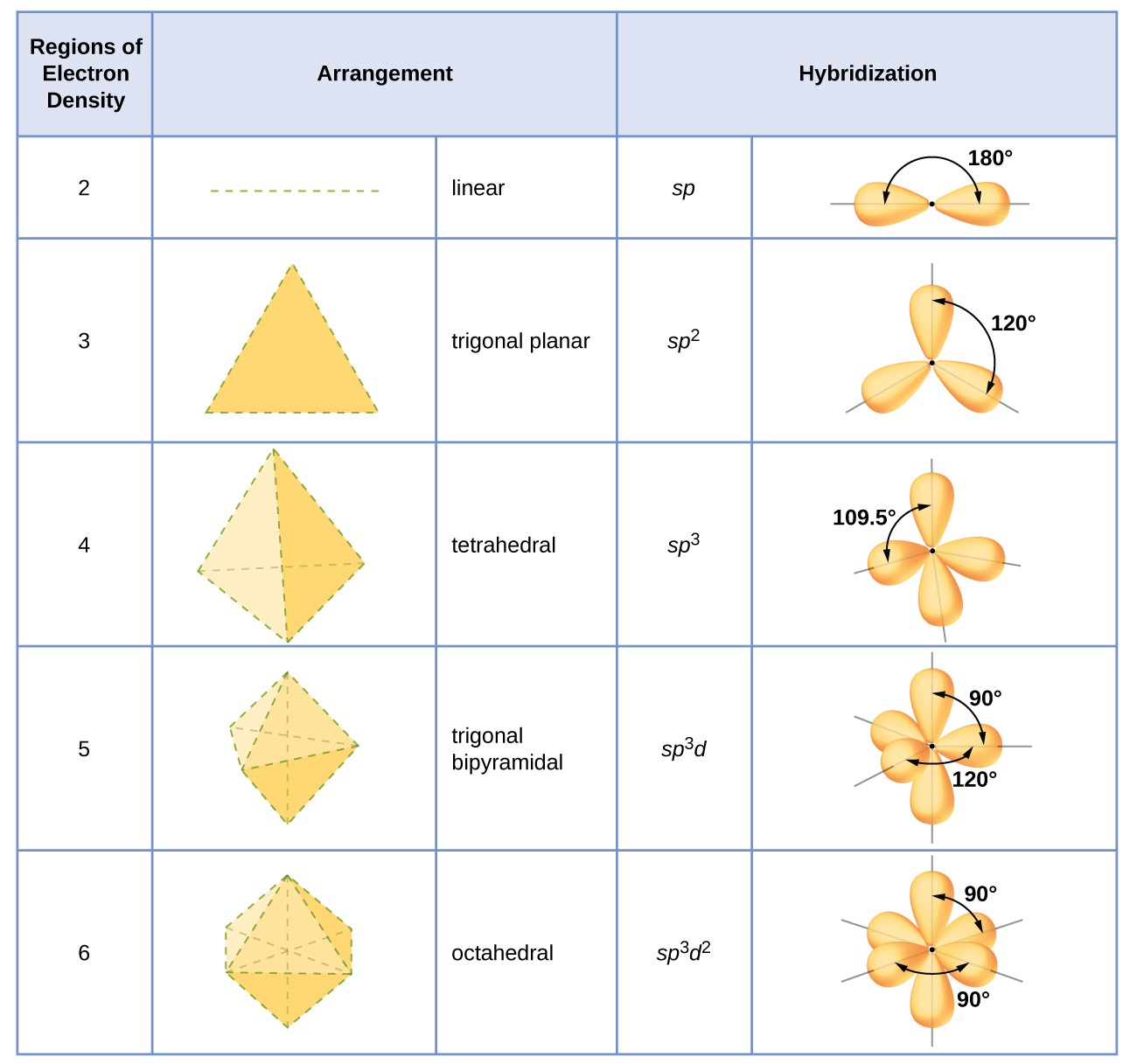
Figure xvi. The shapes of hybridized orbital sets are consistent with the electron-pair geometries. For example, an atom surrounded past three regions of electron density is sptwo hybridized, and the three sptwo orbitals are arranged in a trigonal planar mode.
It is important to remember that hybridization was devised to rationalize experimentally observed molecular geometries. The model works well for molecules containing modest central atoms, in which the valence electron pairs are close together in infinite. Withal, for larger primal atoms, the valence-vanquish electron pairs are further from the nucleus, and there are fewer repulsions. Their compounds exhibit structures that are often not consistent with VSEPR theory, and hybridized orbitals are non necessary to explain the observed data. For example, nosotros take discussed the H–O–H bond angle in H2O, 104.v°, which is more consistent with sp three hybrid orbitals (109.5°) on the fundamental cantlet than with twop orbitals (xc°). Sulfur is in the same group every bit oxygen, and HiiS has a similar Lewis structure. Still, it has a much smaller bond bending (92.1°), which indicates much less hybridization on sulfur than oxygen. Continuing downwards the group, tellurium is even larger than sulfur, and for HtwoTe, the observed bond angle (xc°) is consistent with overlap of the fivep orbitals, without invoking hybridization. Nosotros invoke hybridization where it is necessary to explain the observed structures. 
Example i: Assigning Hybridization
Ammonium sulfate is important every bit a fertilizer. What is the hybridization of the sulfur atom in the sulfate ion, [latex]{\text{So}}_{4}^{2-}[/latex]?
Check Your Learning
What is the hybridization of the selenium atom in SeF4? 
Testify Solution
The selenium cantlet is sp 3 d hybridized.
Instance 2: Assigning Hybridization
Urea, NH2C(O)NHtwo, is sometimes used as a source of nitrogen in fertilizers. What is the hybridization of each nitrogen and carbon atom in urea?
Check Your Learning
Acetic acid, H3CC(O)OH, is the molecule that gives vinegar its odor and sour taste. What is the hybridization of the two carbon atoms in acerb acid?

Prove Solution
H3 C, sp 3;C(O)OH, sp two
Key Concepts and Summary
Nosotros can employ hybrid orbitals, which are mathematical combinations of some or all of the valence atomic orbitals, to describe the electron density effectually covalently bonded atoms. These hybrid orbitals either form sigma ([latex]\sigma[/latex]) bonds directed toward other atoms of the molecule or contain lone pairs of electrons. We tin can decide the type of hybridization around a central atom from the geometry of the regions of electron density about it. Two such regions imply sp hybridization; three, sp 2 hybridization; four, sp 3 hybridization; five, sp 3 d hybridization; and six, sp 3 d ii hybridization. Pi (π) bonds are formed from unhybridized diminutive orbitals (p or d orbitals).
Try Information technology
- Why is the concept of hybridization required in valence bond theory?
- Give the shape that describes each hybrid orbital set:
- sp 2
- sp iii d
- sp
- sp iii d ii
- Explain why a carbon atom cannot form five bonds using sp 3 d hybrid orbitals.
- What is the hybridization of the central atom in each of the following?
- BeH2
- SF6
- [latex]{\text{PO}}_{4}^{\text{iii-}}[/latex]
- PCl5
- A molecule with the formula AB3 could take one of four unlike shapes. Requite the shape and the hybridization of the central A atom for each.
- Methionine, CH3SCH2CHiiCH(NH2)CO2H, is an amino acid constitute in proteins. Draw a Lewis structure of this chemical compound. What is the hybridization blazon of each carbon, oxygen, the nitrogen, and the sulfur?

- Sulfuric acrid is manufactured by a series of reactions represented by the post-obit equations:[latex]{\text{Due south}}_{viii}\left(s\right)+8{\text{O}}_{2}\left(g\right)\rightarrow eight{\text{And then}}_{2}\left(g\correct)[/latex][latex]2\text{South}{\text{O}}_{ii}\left(g\correct)+{\text{O}}_{2}\left(1000\right)\rightarrow 2{\text{And then}}_{3}\left(g\correct)[/latex][latex]{\text{SO}}_{3}\left(thousand\right)+{\text{H}}_{2}\text{O}\left(l\right)\rightarrow{\text{H}}_{2}{\text{So}}_{four}\left(50\right)[/latex] Draw a Lewis structure, predict the molecular geometry by VSEPR, and determine the hybridization of sulfur for the following:
- round S8 molecule
- And thentwo molecule
- SO3 molecule
- HiiSO4 molecule (the hydrogen atoms are bonded to oxygen atoms)
- Ii important industrial chemicals, ethene, CtwoHfour, and propene, C3Hhalf-dozen, are produced by the steam (or thermal) cracking process: [latex]2{\text{C}}_{iii}{\text{H}}_{8}\left(k\right)\rightarrow{\text{C}}_{ii}{\text{H}}_{four}\left(g\right)+{\text{C}}_{3}{\text{H}}_{6}\left(chiliad\right)+{\text{CH}}_{iv}\left(1000\right)+{\text{H}}_{2}\left(g\right)[/latex] For each of the four carbon compounds, do the post-obit:
- Describe a Lewis construction.
- Predict the geometry about the carbon atom.
- Make up one's mind the hybridization of each type of carbon atom.
- For many years after they were discovered, it was believed that the noble gases could not grade compounds. Now we know that belief to exist incorrect. A mixture of xenon and fluorine gases, confined in a quartz bulb and placed on a windowsill, is constitute to slowly produce a white solid. Analysis of the compound indicates that it contains 77.55% Xe and 22.45% F by mass.
- What is the formula of the compound?
- Write a Lewis structure for the compound.
- Predict the shape of the molecules of the compound.
- What hybridization is consequent with the shape yous predicted?
- Consider nitrous acrid, HNOii (HONO).
- Write a Lewis construction.
- What are the electron pair and molecular geometries of the internal oxygen and nitrogen atoms in the HNOii molecule?
- What is the hybridization on the internal oxygen and nitrogen atoms in HNO2?
- Strike-anywhere matches contain a layer of KClO3 and a layer of P4Sthree. The oestrus produced by the friction of hit the friction match causes these 2 compounds to react vigorously, which sets fire to the wooden stem of the match. KClOiii contains the [latex]{\text{ClO}}_{3}^{-}[/latex] ion. P4South3 is an unusual molecule with the skeletal structure.

- Write Lewis structures for P4South3 and the [latex]{\text{ClO}}_{three}^{-}[/latex] ion.
- Depict the geometry nearly the P atoms, the S atom, and the Cl cantlet in these species.
- Assign a hybridization to the P atoms, the S atom, and the Cl atom in these species.
- Determine the oxidation states and formal accuse of the atoms in PfourSthree and the [latex]{\text{ClO}}_{3}^{-}[/latex] ion.
- Place the hybridization of each carbon atom in the post-obit molecule. (The arrangement of atoms is given; you need to decide how many bonds connect each pair of atoms.)

- Write Lewis structures for NF3 and PFv. On the basis of hybrid orbitals, explain the fact that NF3, PF3, and PFfive are stable molecules, but NFfive does not be.
- In addition to NFiii, ii other fluoro derivatives of nitrogen are known: Northward2F4 and N2Ftwo. What shapes practice y'all predict for these two molecules? What is the hybridization for the nitrogen in each molecule?
Glossary
hybrid orbital:orbital created by combining atomic orbitals on a cardinal cantlet
hybridization:model that describes the changes in the atomic orbitals of an atom when information technology forms a covalent chemical compound
sp hybrid orbital:one of a set of two orbitals with a linear arrangement that results from combining one s and one p orbital
sp ii hybrid orbital:one of a ready of three orbitals with a trigonal planar organization that results from combining i s and two p orbitals
sp 3 hybrid orbital:1 of a set of four orbitals with a tetrahedral arrangement that results from combining one south and three p orbitals
sp 3 d hybrid orbital:1 of a set of v orbitals with a trigonal bipyramidal organization that results from combining one s, 3 p, and one d orbital
sp 3 d ii hybrid orbital:i of a set up of 6 orbitals with an octahedral system that results from combining one southward, three p, and 2 d orbitals
reaganhatomentand58.blogspot.com
Source: https://courses.lumenlearning.com/chemistryformajors/chapter/hybrid-atomic-orbitals/
0 Response to "Your Answer Is Incorrect Try Again How Many Sigma Bonds Are Present in the Following Compound"
Post a Comment